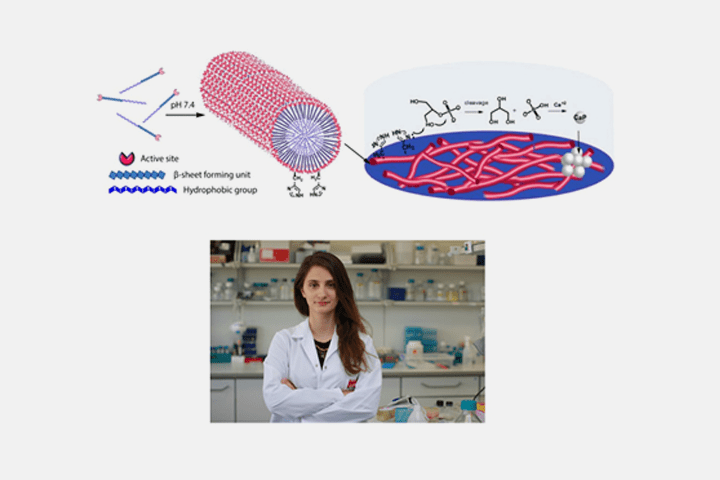
Although produced in abundance by even the simplest of bacteria, the sheer catalytic efficiency of enzymes cannot be matched by any chemical catalyst – human effort cannot yet replicate what has been optimized through aeons of natural selection. The imitation of enzyme structure and function is therefore an important concern for the development of new catalysts, and peptide amphiphiles, with their amino acid-based compositions, are ideal materials for this purpose. Owing to the structural complexity of enzymes, the peptide-mediated mimicry of enzymatic function has been uncommon in the literature, but the groups of Drs. Ayşe Begüm Tekinay and Mustafa Özgür Güler have now announced the successful imitation of a particularly complex enzyme – alkaline phosphatase (ALP), a multi-functional catalyst that both regulates and directly facilitates the formation of bone.
The primary function of alkaline phosphatase is to detach phosphate groups from various organic molecules, which occurs through the activity of histidine amino acids present in its structure. Indeed, these histidines are so vital that they can evidently function in the absence of the rest of the enzyme – the peptide networks developed by the team are little more than a series of histidines in a well-coordinated, one-dimensional array, but nonetheless display a formidable ability to cleave phosphates from an organic substrate. While ALP is found ubiquitously in the human body, it is especially abundant in bone, where the phosphate groups it produces are channeled to the tissue biomineralization process – and ALP-mimetic peptide networks are also capable of replicating this function, driving preosteoblast cells to differentiate into mature cells and start forming bone nodule-like structures.
In addition to serving as a potent means of inducing osteoblast differentiation and in vitro biomineralization, the peptide material can also function as a model for the study of enzymatic activity, allowing the investigation of both the catalytic and metabolic functions of ALP. The authors are especially hopeful about the biomedical applications of the material, as the scaffold material is biodegradable and similar in composition to the native extracellular matrix of bone – properties that cannot be replicated by metal and ceramic implants.
Their work has been published in the journal Biomacromolecules and can be accessed at the following address: